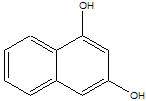
The structure at right shows the molecule called naphthoresorcinol. It is unusual because it it has a large planar aromatic surface (like naphthalene) but it also has two OH groups, making it a diol. It turns out some amino acids, when they are linked together in sequence, can orient their carbonyl (C=O) groups almost perfectly such they they point to the hydrogen atoms of the OH groups on naphthoresorcinol if the molecules are brought close together. In addition, the amino acids are partially preorganized, which means they have many fewer conformations available to them, essentially forming a rigid binding site for the diol.
_________________________________________________________
_________________________________________________________